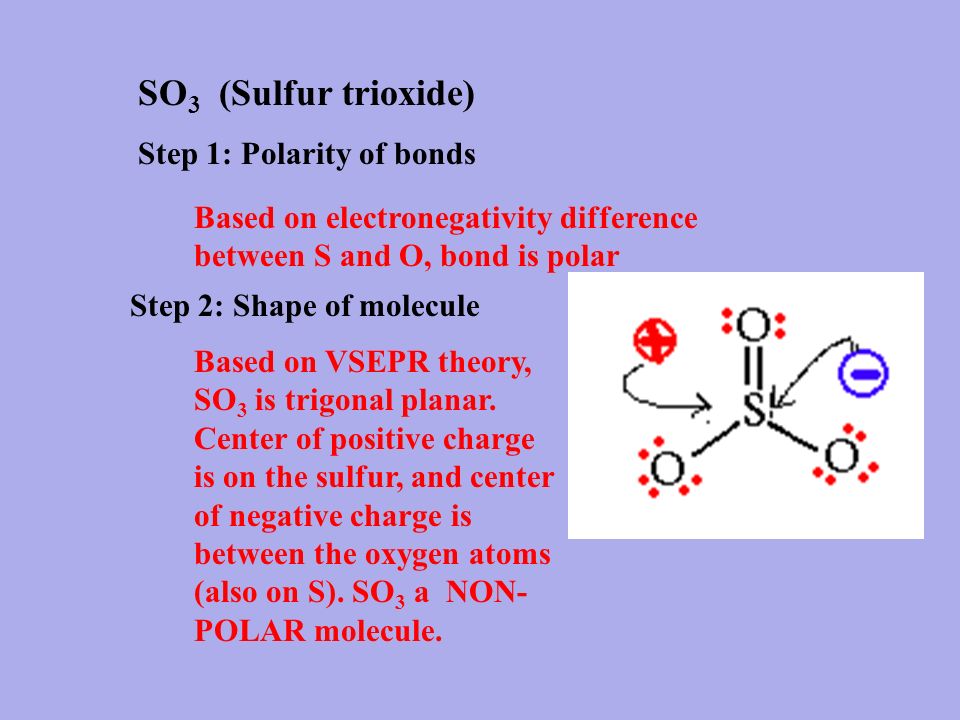
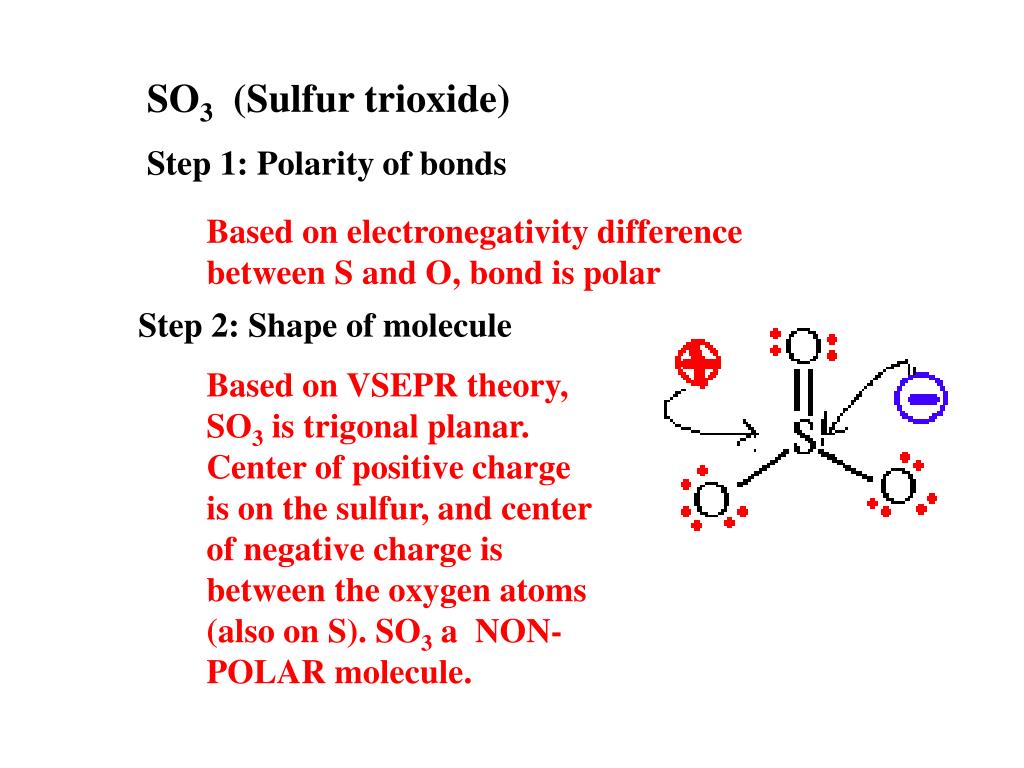
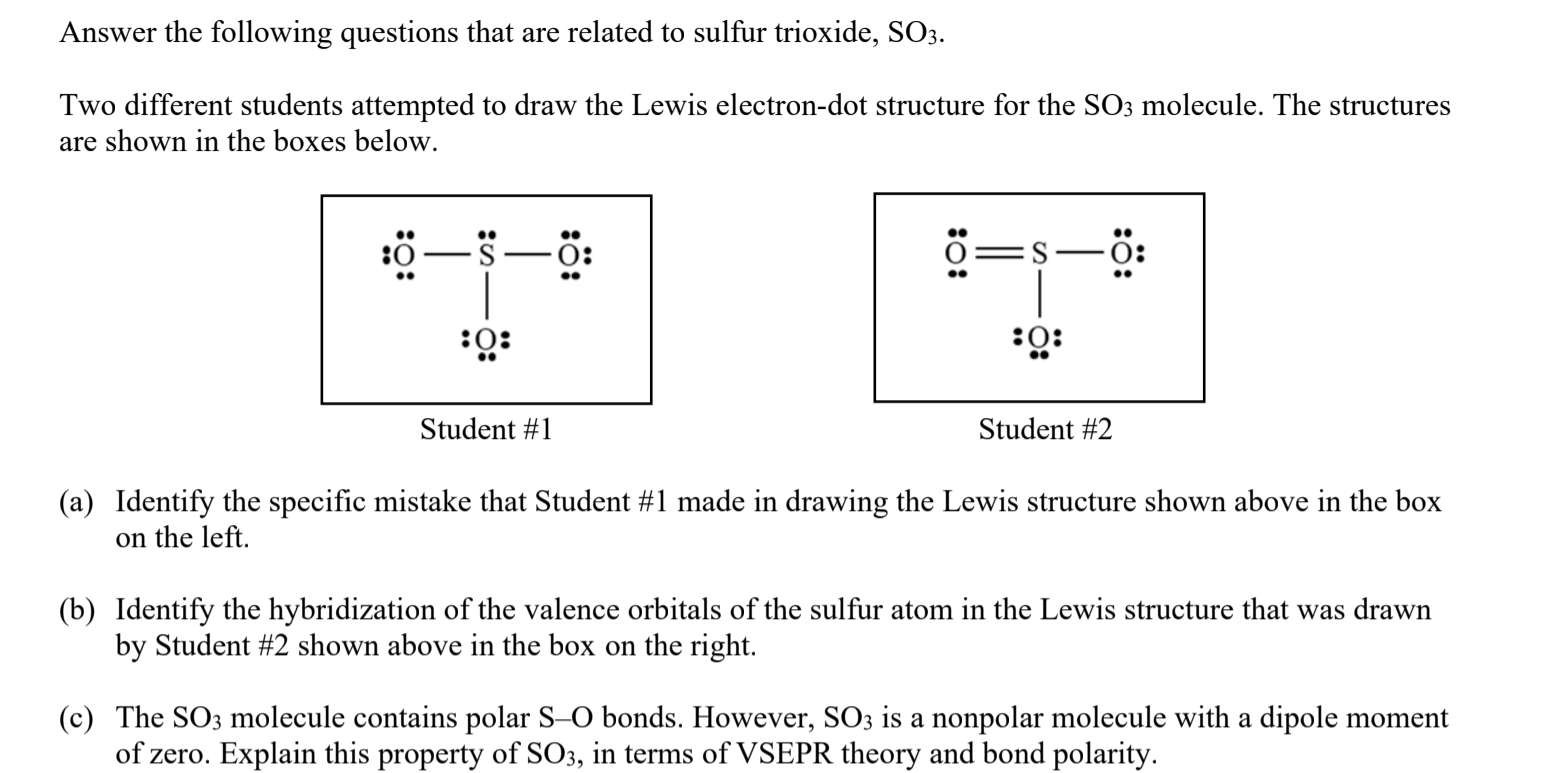
amongst NO3^ 1, AsO3^ 3,CO3^ 2, ClO3^ 1, SO3^ 2 and BO3^ 3, the non polar species are: (1) NO3^ 1,CO3^ 2, BO3^ 3 (2) AsO3^ 3, ClO3^ 1, SO3^ 2 (3) CO3^ 2, SO3^ 2, AsO3 3 (4) ClO3^ 1, SO3^ 2, NO3^ 1

SO3 Lewis Structure - How to Draw the Dot Structure for SO3 | Lewis Structure| Chemical Bonding | Success in Chemistry

Draw the electron-dot structure for SO3. What is its molecular shape? Is it polar or nonpolar? | Homework.Study.com
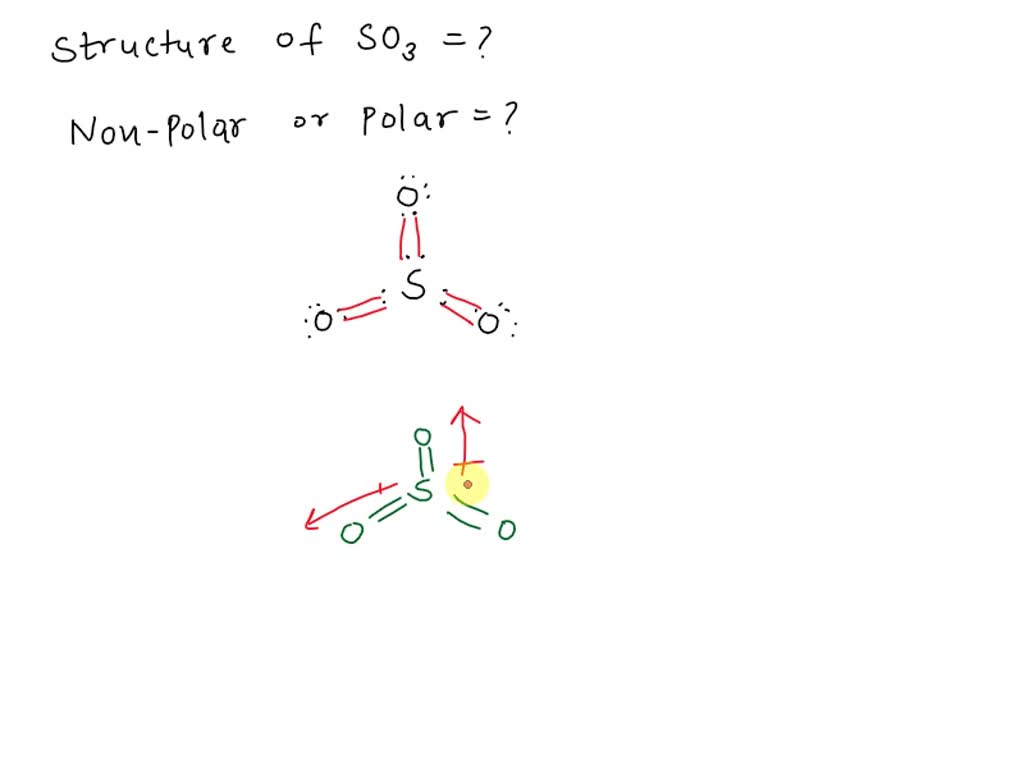
SOLVED: SO3 is a covalent compound with no lone pairs on the central S atom. What's the structure of a molecule of SO3? Is an SO3 molecule polar or non-polar? Question 14
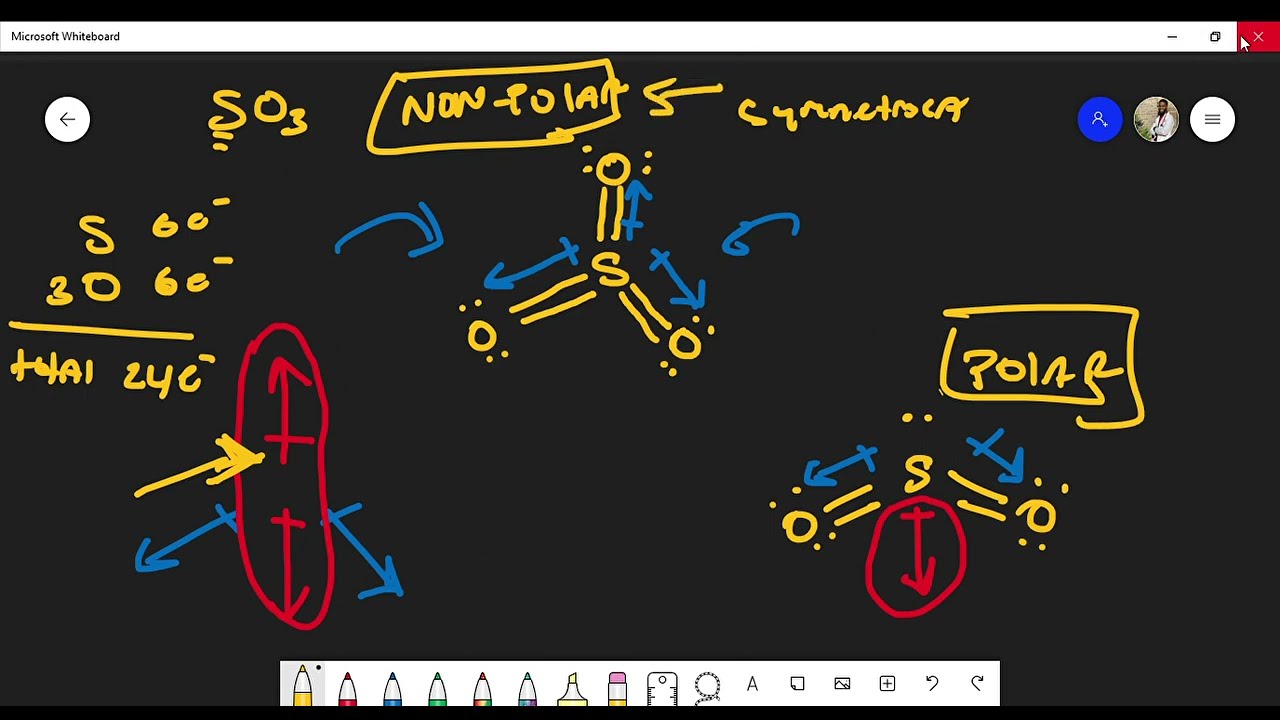
Is Sulfur Trioxide (SO3) Polar or Non-Polar? Lewis Structure (The Difference Between SO2 and SO3) - YouTube

Draw the Lewis structure for SO3. Determine the number of electron groups, the electron geometry, and the molecular shape. Is it polar or nonpolar? | Homework.Study.com

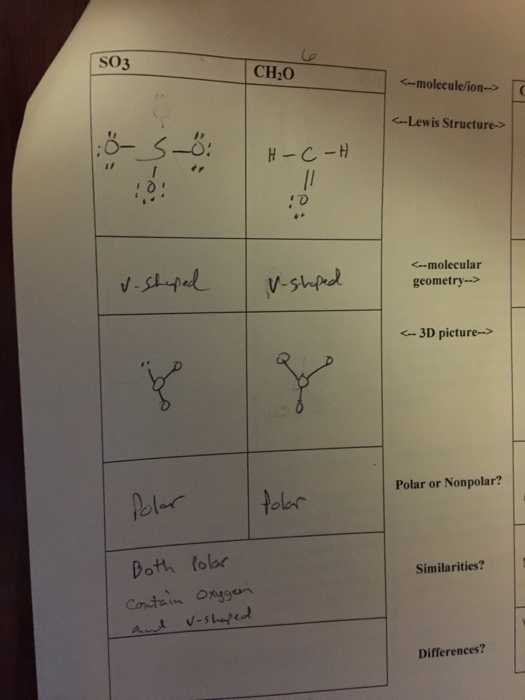
Depicted below you will see the molecules COH2 and SO3 . Why is COH2 polar while SO3 isn't? - brainly.com
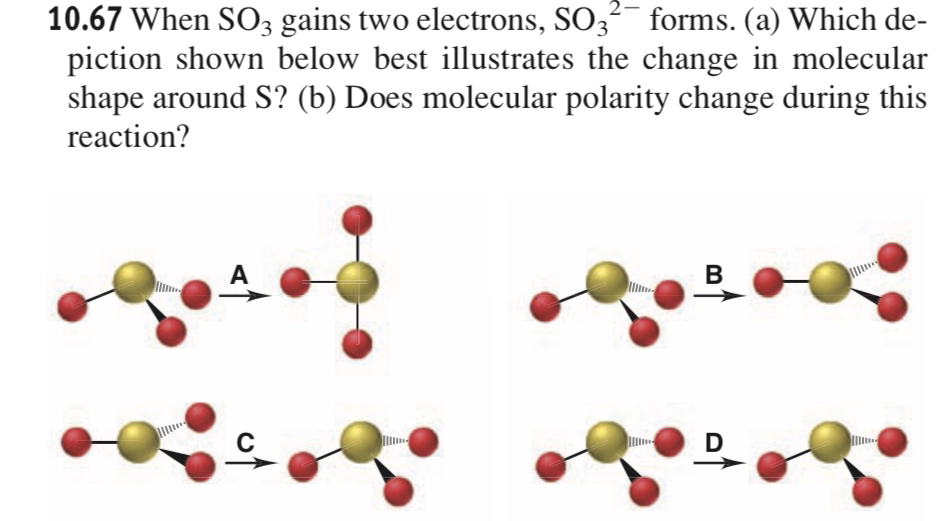
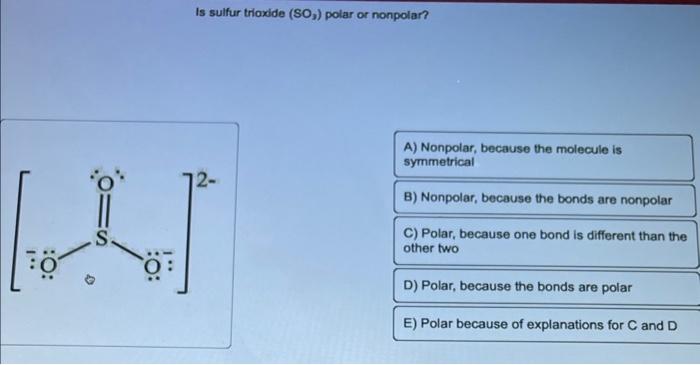
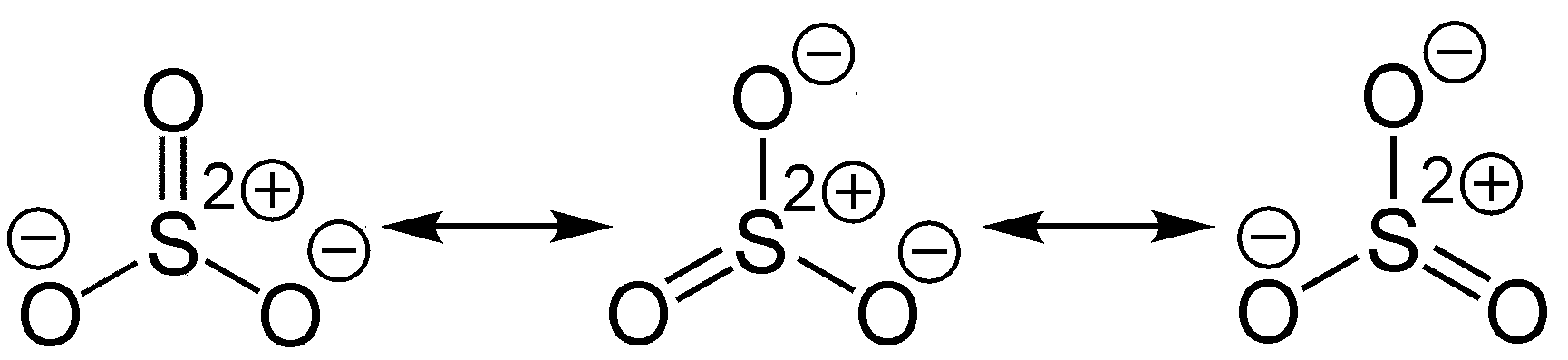
SOLVED: 10.67 When SO3 gains two electrons, SO3^2- forms. (a) Which depiction shown below best illustrates the change in molecular shape around S? (b) Does molecular polarity change during this reaction?

Which of the following molecules has polar bonds but is a nonpolar molecule? a. CO b. Cl2 c. SO3 d. HBr e. none of the above | Homework.Study.com