Is CHCl3 Polar or Nonpolar? - Polarity of Chloroform | Molecular geometry, Covalent bonding, Chloroform
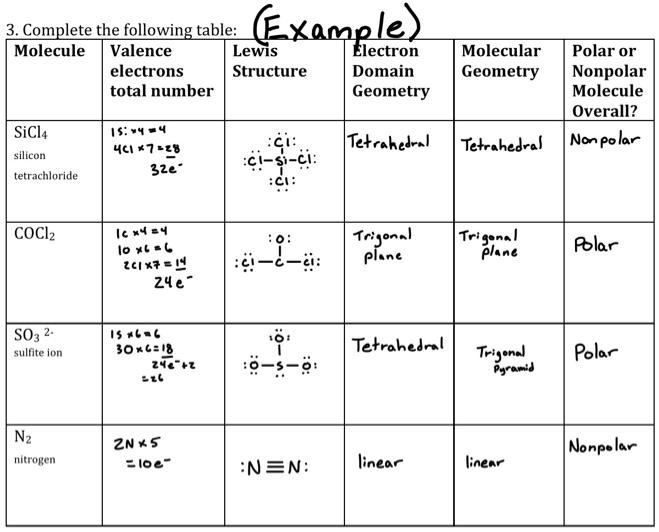
SOLVED: Complete the following table: Molecule Valence Lewis Electrons Structure Domain Total Number Geometry Molecular Geometry Polar or Nonpolar Molecule Overall? Nonpolar SiCl4 Silicon Tetrachloride 4 7-2-4 3-2-4 Tetrahedral Tetrahedral Nonpolar COCl2

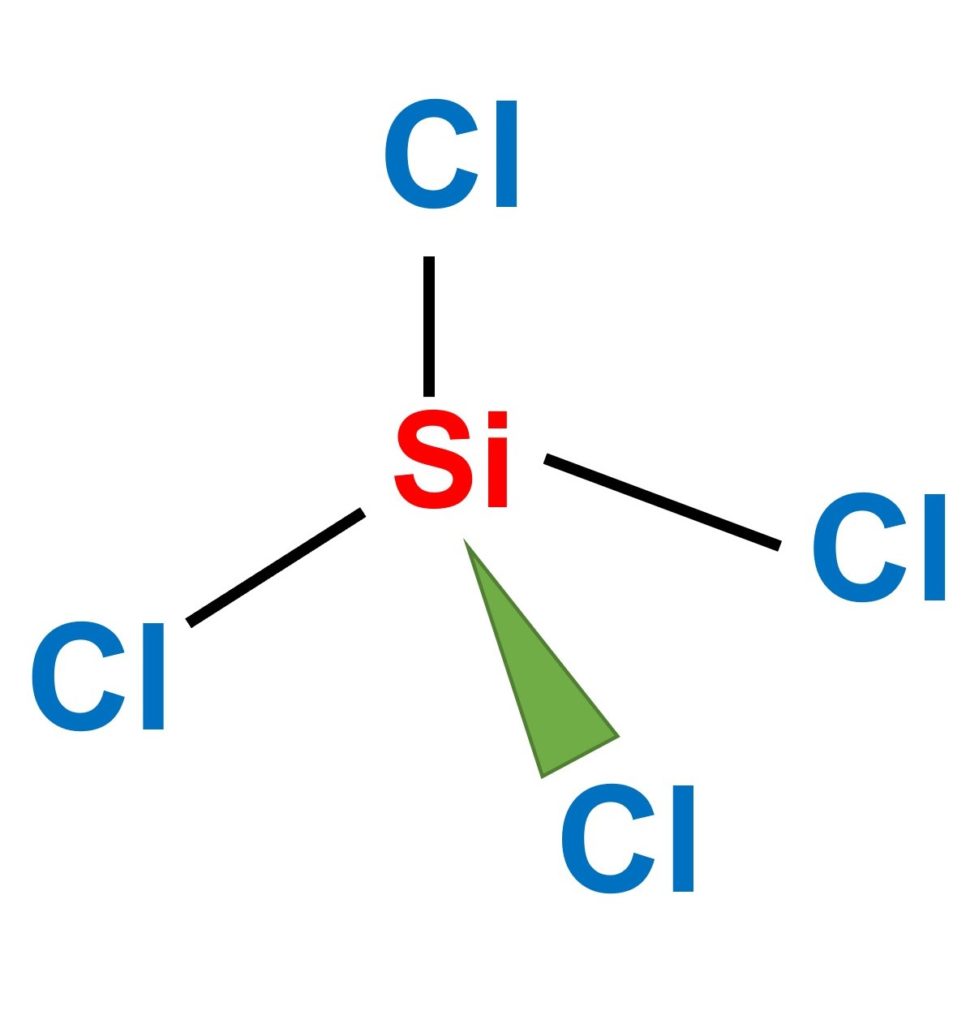
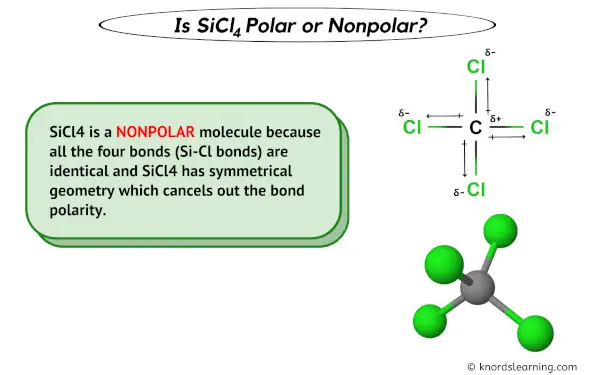
Is SiCl4 Polar or Non-polar? (Silicon Tetrachloride) | Is SiCl4 Polar or Non-polar? (Silicon Tetrachloride) We know the concept of polarity is a bit tricky and one might need a little practice
![The \\[SiC{l_4}\\] molecule is nonpolar and chlorine is more electronegative than silicon. From this information alone it can be deduced that:1. \\[Si-Cl\\] bond is nonpolar2. \\[SiC{l_4}\\] molecule is planar3. \\[SiC{l_4}\\] molecule is The \\[SiC{l_4}\\] molecule is nonpolar and chlorine is more electronegative than silicon. From this information alone it can be deduced that:1. \\[Si-Cl\\] bond is nonpolar2. \\[SiC{l_4}\\] molecule is planar3. \\[SiC{l_4}\\] molecule is](https://www.vedantu.com/question-sets/cff3a96a-22a9-491b-b4c7-e2a25d03a8828875529946816540776.png)
The \\[SiC{l_4}\\] molecule is nonpolar and chlorine is more electronegative than silicon. From this information alone it can be deduced that:1. \\[Si-Cl\\] bond is nonpolar2. \\[SiC{l_4}\\] molecule is planar3. \\[SiC{l_4}\\] molecule is

Arrange the following molecules from most to least polar: CH4, CF2Cl2, CF2H2, CCl4, and CCl2H2. | Homework.Study.com
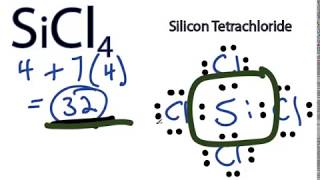
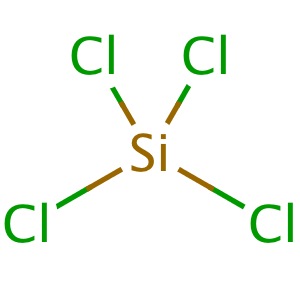
SiCl4 Lewis Structure: How to Draw the Dot Structure for SiCl4 | Chemical Bonding | Success in Chemistry