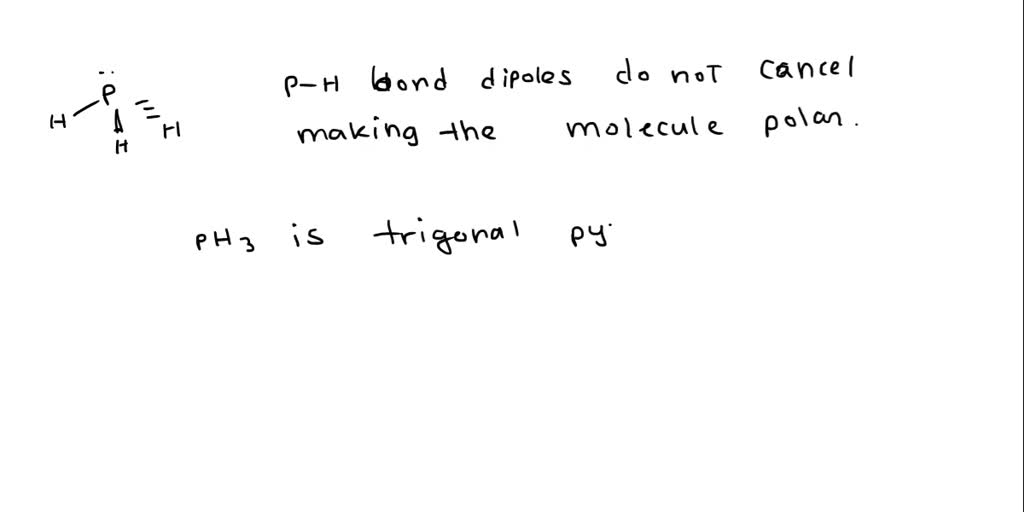a) Optimized structures of the key phosphoranyl radicals. Non-polar... | Download Scientific Diagram

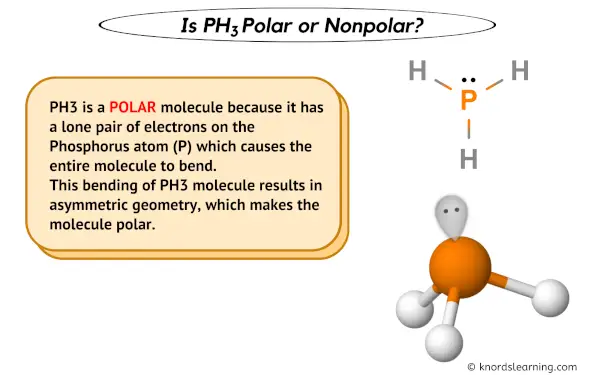
Draw the Lewis structure for PH3. How many bonds and non-bonding pairs are around the central atom, and what is the shape of this molecule? | Homework.Study.com