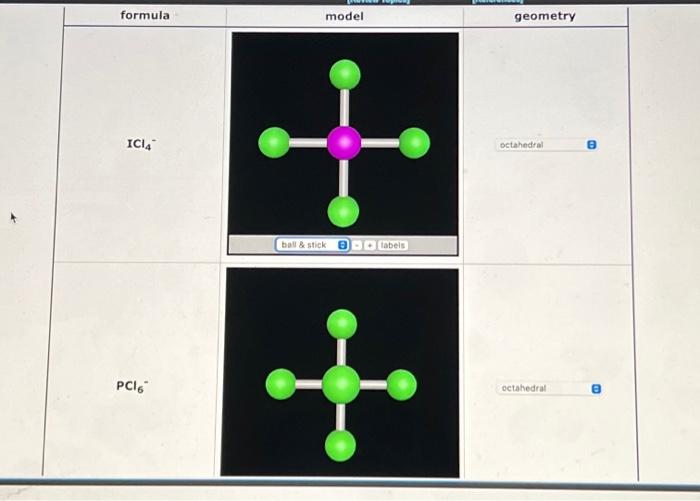
Geometry of Molecules - Geometry of Molecules ABn 0 Lone Pairs n=2 1 Lone Pairs 2 Lone Pairs BAB A A linear angle: 180 Ex: CO2 BeCl2 Hybridization: | Course Hero

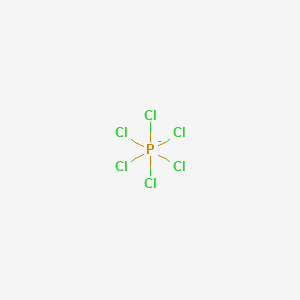
Devise the Lewis formula, sketch the three-dimensional shape, and name the electronic and ionic geometries for the given ion. PCl6 - | Homework.Study.com

Mooer PCL6 MKII Pedal Controller Programmable Loopswitcher with 6 Loops L6 PL6 With Free Connector Guitar Accessories - AliExpress
![VSEPR theory working out shapes of molecules ions deducing bond angles linear trigonal planar pyramid bypyramid tetrahedral octahedral T shape electron pair molecular geometry BeH2 BeCl2 CO2 [Ag(NH3)2]+ BH3 BF3 BCl3 AlF3 VSEPR theory working out shapes of molecules ions deducing bond angles linear trigonal planar pyramid bypyramid tetrahedral octahedral T shape electron pair molecular geometry BeH2 BeCl2 CO2 [Ag(NH3)2]+ BH3 BF3 BCl3 AlF3](https://www.docbrown.info/page06/addhoc06/pcl6-OX.gif)
VSEPR theory working out shapes of molecules ions deducing bond angles linear trigonal planar pyramid bypyramid tetrahedral octahedral T shape electron pair molecular geometry BeH2 BeCl2 CO2 [Ag(NH3)2]+ BH3 BF3 BCl3 AlF3



![What is the molecular structure of [PCl_6]^-, and what is the bond angle? | Homework.Study.com What is the molecular structure of [PCl_6]^-, and what is the bond angle? | Homework.Study.com](https://homework.study.com/cimages/multimages/16/pcl62958494712247179425.png)