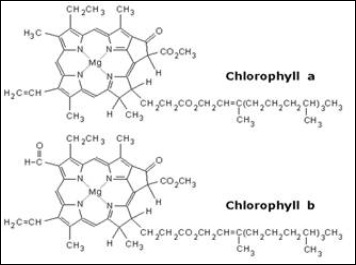
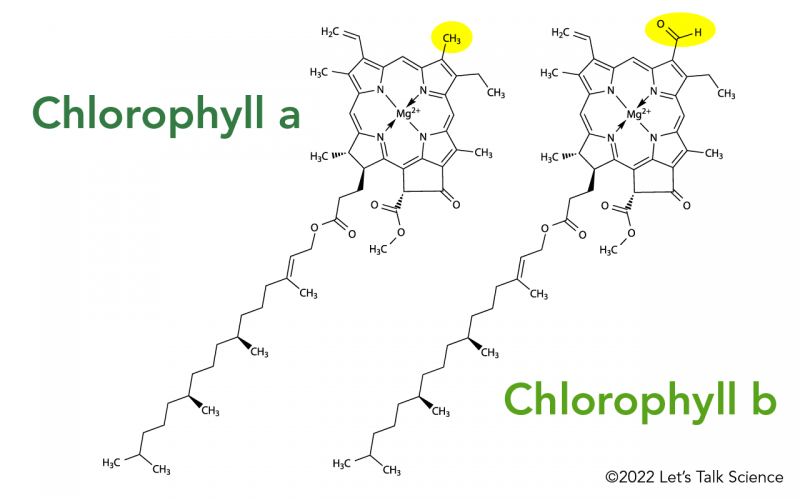
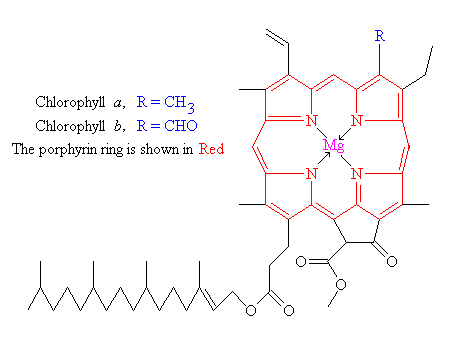
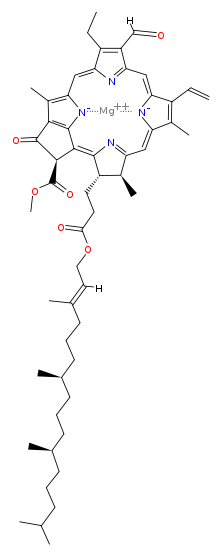
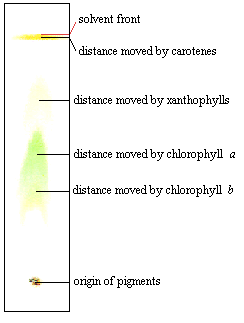
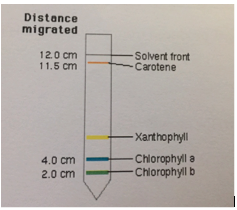
What is the chemical identity of the spots on the TLC plate for a spinach extract? There are 4 spots (chlorophyll A, Chlorophyll B, Phenophytin, Xathophylls). Explain the reasoning behind your assign

Isolation of Chlorophyll and Carotenoid Pigments from Spinach Pages 1-7 - Flip PDF Download | FlipHTML5

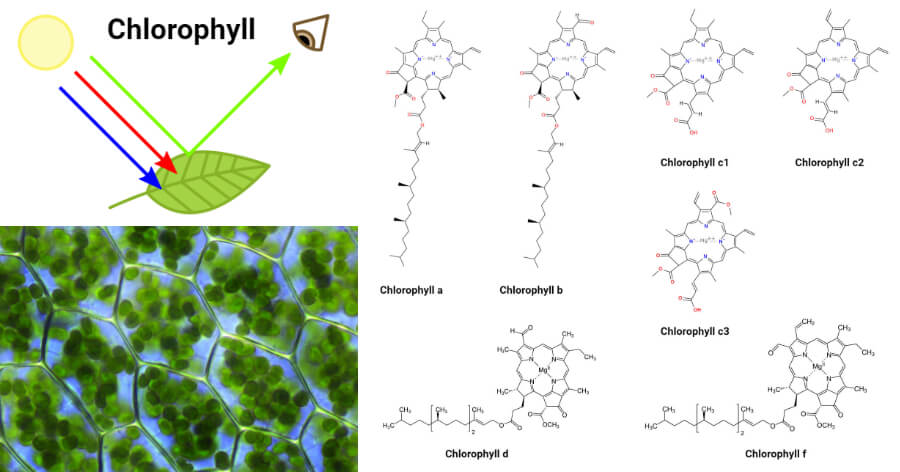
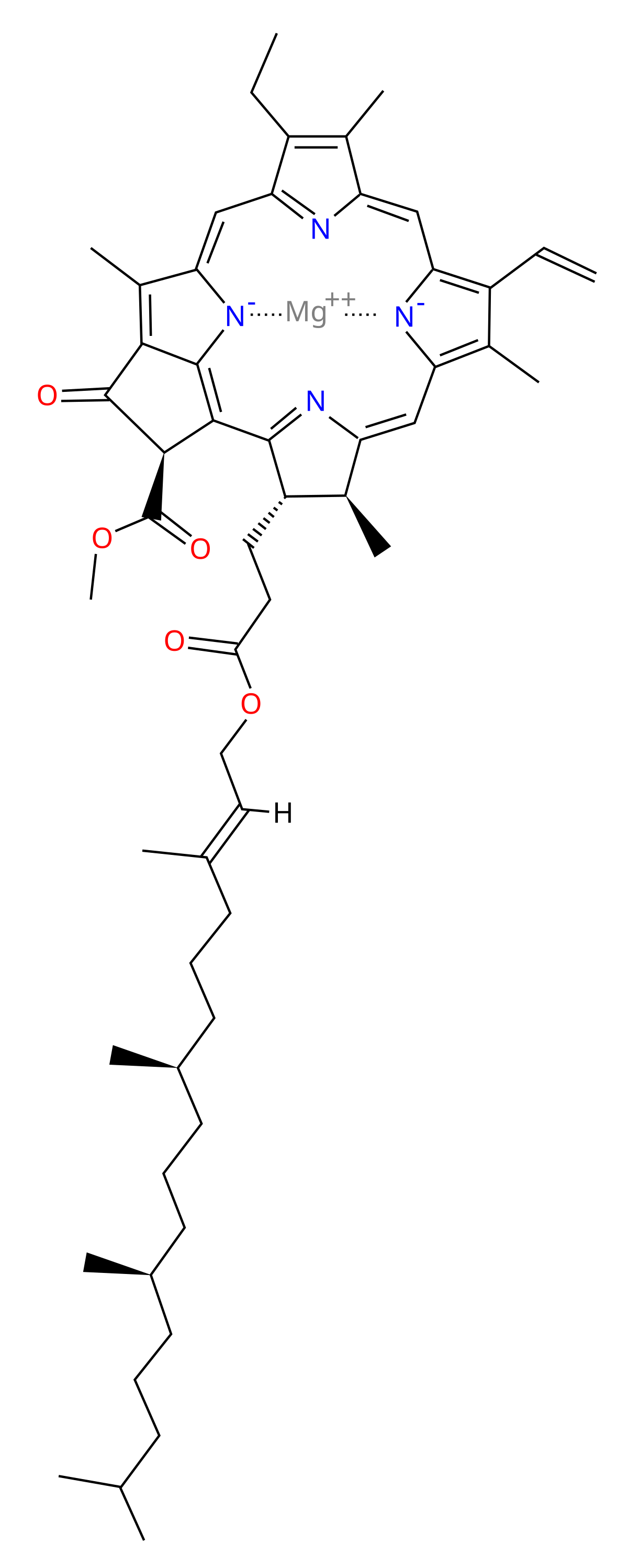
The chemical structure of typical chlorophyll molecule. Chlorophyll a... | Download Scientific Diagram
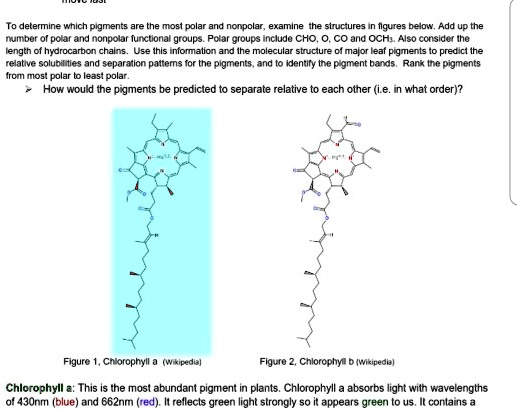
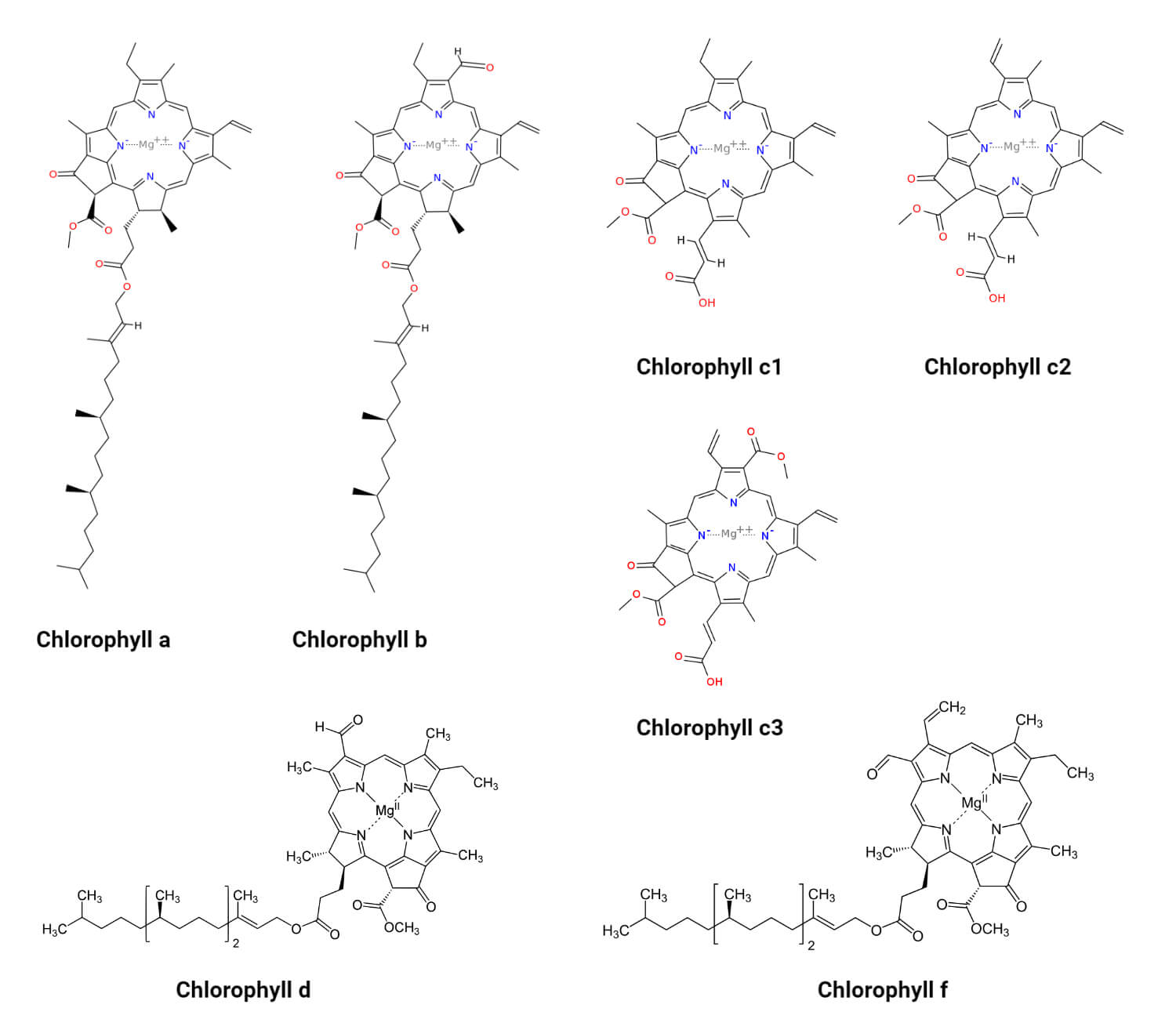
SOLVED: To determine which pigments are the most polar and nonpolar, examine the structure figures below: Add up the number of polar and nonpolar functional groups. Polar groups include CHO, CO, and

Dependence of the chlorophyll wavelength on the orientation of a charged group: Why does the accessory chlorophyll have a low site energy in photosystem II? - ScienceDirect

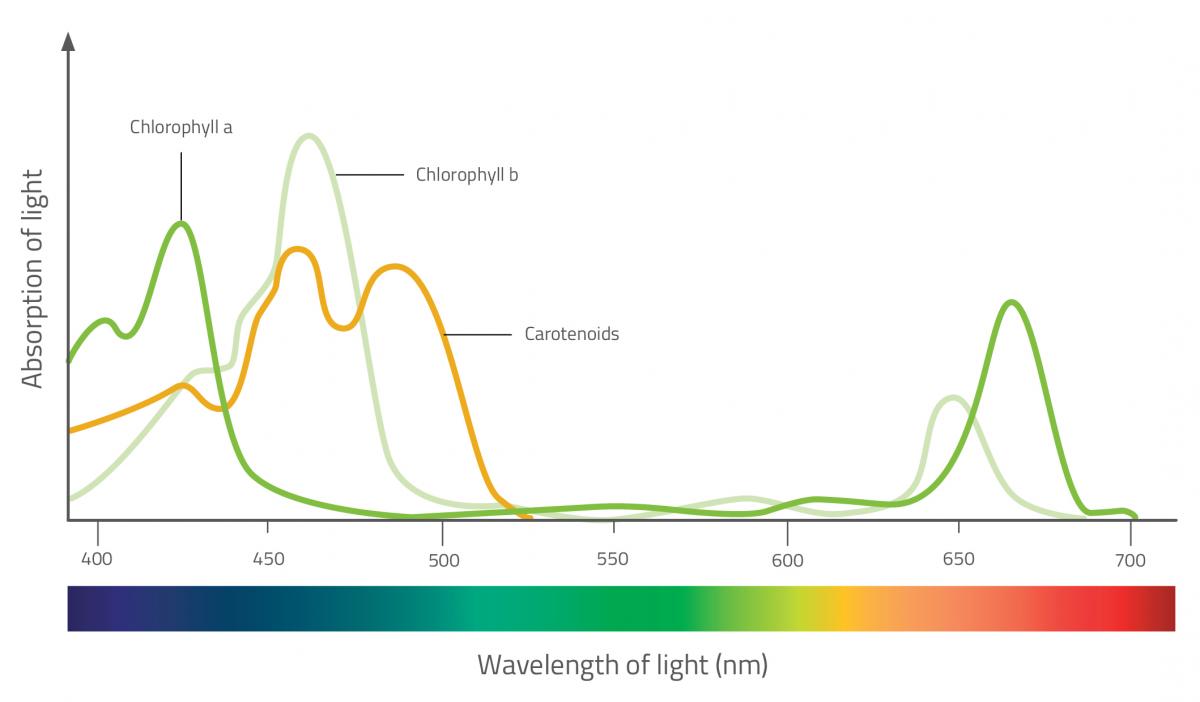
![생명과학/실험] Separation and Identification of Plant Pigments by Paper Chromatography 생명과학/실험] Separation and Identification of Plant Pigments by Paper Chromatography](https://t1.daumcdn.net/cfile/tistory/20780B3A4D9E94480C)